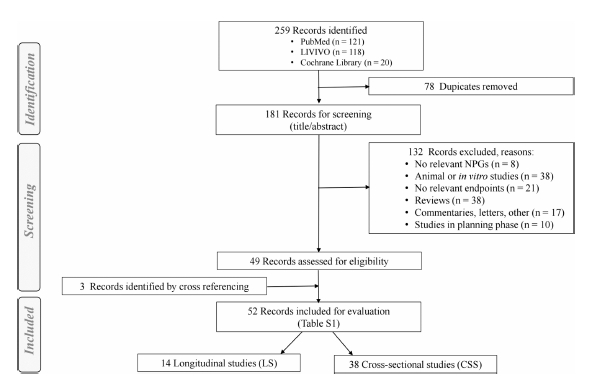
Review results
We performed a qualitative synthesis of the available evidence regarding oral health risks for users of electronic cigarettes (ECs) and oral nicotine products.
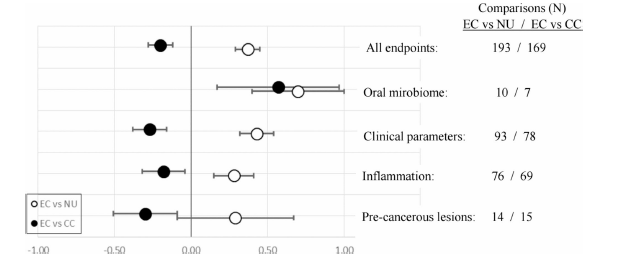
The review discusses the results from 52 studies suggesting a reduced risk for EC users as compared to smoking. Yet, oral health status remains poorer compared to not using any tobacco/nicotine products. This is indicated by the scores in the following figure vor EC usage. Negative score illustrates potentially reduced risk for people who vape compared to people who smoke, which is generally the case. However, there is still an elevated risk relative to no nicotine/tobacco use. Most studies lack comprehensive characterization of cohorts and compliance monitoring
There are major gaps in most of the studies, unfortunately.
We encourage researchers to address those when designing their trials:
Long-term studies are lacking: As e-cigarettes have only been on the market since around 2007, there are no long-term studies to date that could investigate the effects on the development of diseases such as cancer. Most studies have covered a relatively short period of e-cigarette use (usually only a few years). Long-term studies are needed to understand the chronic effects.
Taking into account the variety of ECs: The variety of e-cigarette types, generations, nicotine levels and added flavors is not differentiated in most studies, which contributes to further heterogeneity.
Insufficient consideration of dual use: Many studies do not sufficiently consider the problem of dual use or the previous consumption of tobacco cigarettes. This is a must for an accurate characterization of the sample. Missing this out leads to a bias in the results, as the observed effects may not be exclusively attributable to the use of e-cigarettes. It is important to distinguish between people who use e-cigarettes exclusively and people who also use tobacco cigarettes.
Lack of biomarker validation: There are few studies that use biomarkers to examine exposure to e-cigarette constituents. Many studies rely on self-reporting, which can be prone to error. There is also a lack of long-term biomarkers for tobacco use.
Specific endpoints: There are a variety of endpoints used in studies, making comparability difficult. There is a need to define consistent and validated endpoints to better assess the impact of e-cigarettes on oral health.
Oral microbiome research: The effects of e-cigarettes on the oral microbiome are an important area of research, as changes here are associated with inflammation and other oral problems.
Comparison with other products: Studies comparing the effects of e-cigarettes with other nicotine products such as heated tobacco products (HTPs) or nicotine pouches (ONPs) are scarce. Future studies should pay more attention to these product categories, especially ONPs, as they have intensive contact with the oral mucosa. Close the data gap for nicotine pouches
ECs is associated with relatively fewer detrimental oral health effects compared to smoking, yet oral health status remains poorer compared to not using any tobacco/nicotine products. These results have to be interpreted with caution due to a number of limitations and uncertainties in the
underlying studies, particularly the potential biases and confounding factors inherent in cross-sectional study designs.
Chances for tobacco harm reduction
South Asia has a high use prevalence of toxic smokeless tobacco with drastic impacts on oral health. The opportunity to improve the situation there should not be missed: With quality data derived from the regions with high prevalence.
You can find the full article on our publication page: